The neurosurgical team of Mekelle University’s Ayder Hospital struggles to save the life of a poor farmer from a deadly brain tumor. A critically ill patient who faced death unless he underwent emergent brain surgery. An intense multi-hour confrontation with a brain tumor. This became an almost everyday occurrence for our team.
I had come to Ayder Comprehensive Specialized Hospital in Mekelle, Ethiopia in 2015 to start a neurosurgery program. This was a federal teaching teaching hospital and part of a university medical center serving about 15 million people. We wanted to create a sustainable medical service that also train neurosurgeons.
As the family said goodbye to him he left the ICU for the operating room. He had been intubated and resuscitated in the emergency room where he presented with coma. A young uneducated farmer from a remote primitive area who had been deteriorating for a year and half. Tumors in this part of the world are large and angry because they present so late.
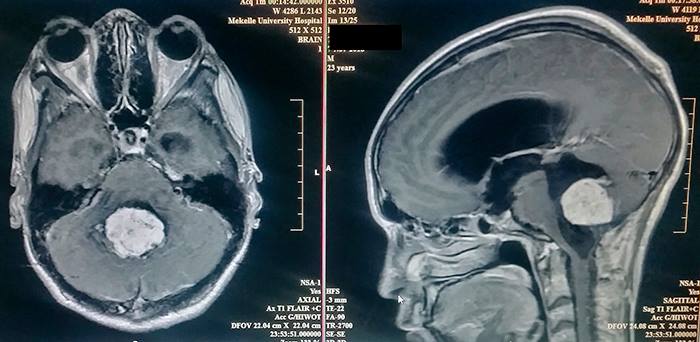
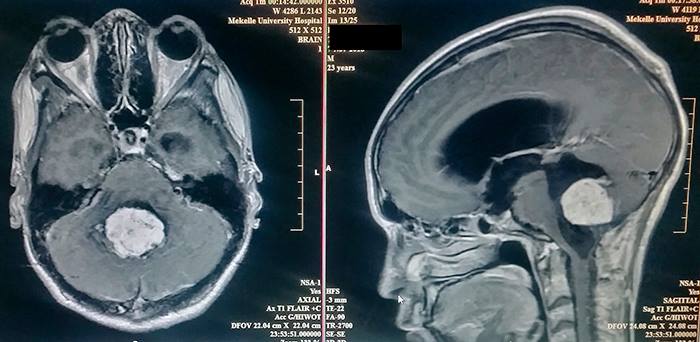
The MRI of the brain showed a 10 centimeter ependymoma in the fourth ventricle which was highly vascularized. This is the type of case neurosurgeons dream of, a mix of danger and hope.
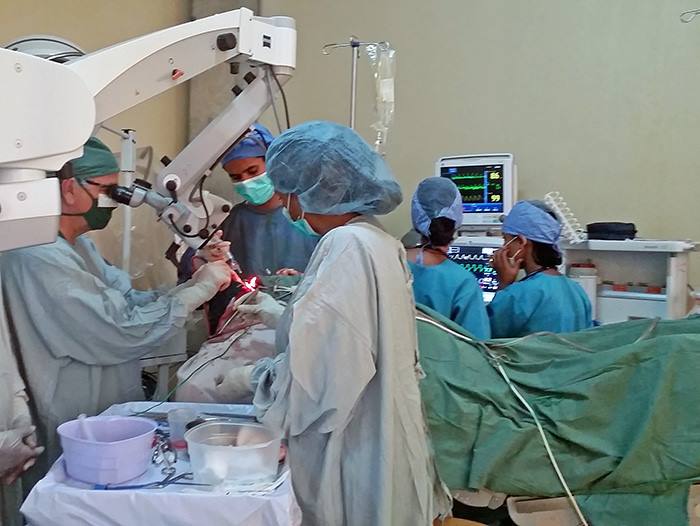
The residents and fellows do the initial opening until the back of the brain is exposed which takes the first hour of surgery. I can see the tumor erupting to surface with very swollen blood vessels around it. Instead of the ependymoma (an easier tumor to remove) this was going to be hard. I begin working a millimeter at a time. Gently dissecting tumor from brain, isolating and separating scar bands and blood vessels. Using directed light and magnification for the next 8 hours I cannot take even a 5 minute break. Removing the tumor means there is constant blood loss. The brain receives more blood than any other organ. Tumors cause extra blood vessels to grow into them. As you control one bleeder another 2 start.
By the fourth hour into surgery he has lost 5 liters of blood and his blood pressure is fragile. By the sixth hours there is still about 20% of the tumor left which is covering the cerebral aqueduct. This must be removed to allow flow of cerebrospinal fluid. We are millimeters away from vital areas that cannot be damaged.
Another 2 hours goes by and there are times when the blood loss starts again and pressure gets low even with another 2 units. After 8 hours from the beginning of my part of the surgery the tumor is completely removed. The patients blood pressure is holding thanks to high dose epinephrine (a drug to raise blood pressure in critical patient). Now comes the hardest time. Will the patient wake up? He goes to the intensive care unit and we wait. After a few hours he begins to move in a way that suggest he will emerge from coma.
This is the life of neurosurgeon. Fighting to stay on the brink of life for hours at a time.